INTRODUCTION The vestibular evoked myogenic potential (VEMP) consists of a vestibulo-neck reflex, middle latency, resulting from acoustic stimulation, a high intensity of the saccule (1-3). The electromyographic activity of neck muscles, particularly the sternocleidomastoid muscle, is captured by means of surface electrodes (4).
The vestibulocervical reflex originates in the saccule, is transmitted to the neurons of Scarpa's ganglion, walks into the inferior vestibular nerve, vestibular nucleus and vestibulospinal tract and reaches the motor neurons of the sternocleidomastoid muscle (5-7).
This potential is investigating whether the vestibular organ is hypersensitive to sound (caused by disease of the inner ear) and whether there are changes in the saccule, inferior vestibular nerve or vestibular nucleus (8-14).
Currently, despite the increase in studies on VEMP (2), the work which are developed with an adapted version, which means limitations on the investigation of the parameters of the test (15-28). The development of an instrument for the assessment of vestibular evoked myogenic potentials with specific amplification, filtering, stimulation and capture window, will allow a thorough adjustment and further research on the subject, which, with a device adapted to assesses auditory evoked potentials, it is not possible.
Thus, this study aimed to compare, through a pilot test, the latencies and amplitudes of the components p13 and n23, and asymmetry indices obtained in the acquisition of vestibular evoked myogenic potential between equipment considered a gold standard, adapted to record the potential, and equipment developed for specific purposes of evaluating the VEMP.
METHOD The protocol of this research is based on relevant legislation, Resolution No. 196/96 of the National Health Council, Ministry of Health for studies in humans and was approved by the Ethics Committee of the institution where the research was conducted, with Protocol 625, on December 1, 2006.
The sample consisted of 11 individuals, six females and five males, spontaneous demand of Acoustics and Instrumentation Laboratory - LIA, considering the following inclusion criteria: ages between 18 and 35 years and hearing thresholds equal or below 20 dB HL with differences between the ears, often have less than 10 dB. The exclusion criteria adopted were: Exposure to occupational or recreational noise, ear surgeries, more than three ear infections in the current year, use of ototoxic medication, tinnitus, vertigo, dizziness or other cochlear-vestibular changes, presence of systemic changes that may contribute to cochlear-vestibular disorders such as diabetes, hypertension and dyslipidemia and / or hormonal changes.
Initially, we applied a screening questionnaire, then the research was explained verbally and a term of informed consent has been delivered and signed before the beginning of the experiment the subjects who agreed to participate.
Before the capture of the VEMP were performed the following procedures to verify the normality of hearing: a) Otoscopy for inspection of the external auditory canal and the integrity of the tympanic membrane with an otoscope of Welch Allyn 29090 with sterilized speculums. b) Pure tone audiometry with audiometer model AC40 Interacoustic brand, pure tone pulsed cabin acoustics.
The vestibular evoked myogenic potential (VEMP) was recorded in two facilities, equipment gold standard (the Interacoustic EP25 - widely used instrument for assessing potential short, medium and long latencies) and the apparatus developed.
The new device was developed at the Laboratory of Acoustics and Instrumentation (LIA) of UNCISAL and Instrumentation Centre of Radiation Protection Dosimetry and FFCLRP-USP (CIDER), which is composed of biological amplifiers, with gain up 200.000x, bandpass filters configurable between 0 10,000 Hz, electrical protection system and a logical system that allows detailed investigation of VEMP, since it allows for broad control of the parameters of the test, different models available that have pre-defined parameters. Moreover, the device has an interface with the computer via the sound card, which can decrease its value to trade at around 50% compared to the models currently available. In both devices, the sound stimuli were presented through earphones ER-3A insert.
Initially, the volunteers sat in a chair and were cleaned from the skin thereof, using abrasive paste, alcohol and cotton. Then, disposable electrodes were placed on the type silver and silver chloride (Ag / AgCl) for potential recording, after previous use of electrolytic paste. The active electrode was positioned in the upper half of the sternocleidomastoid muscle, ipsilateral to stimulation, the reference electrode over the anterior border of the clavicle, ipsilateral, and the ground electrode in the frontal midline.
After placement of the electrodes, we preceded with the evaluation of the impedance between the electrodes non-inverting and ground electrodes and between the inverter and ground. Thus, it was allowed impedance between the electrodes of up to 3 k W.
To obtain registration of the vestibular evoked myogenic potentials, sternocleidomastoid muscle, the patient remained sitting, with maximum lateral rotation of head to the side contralateral to stimulation and maintained tonic contraction of the muscle around 60 microvolts. The stimulus was initiated by afferent right and then repeated in the afferent left. The responses were replicated, ie recorded twice on the right side and twice on the left.
In evoking VEMP by equipment EP25, 200 stimuli were averaged type tone burst, lasting 10 ms (rise: 4, Plateau: 2, descent, 4), the frequency of 500 Hz, presented at a rate of 5 Hz, with an intensity of 90 dB Nan, using a bandpass filter 5-1200 Hz records were performed in windows of 80 ms and a channel registration since the capture of the stimuli and the evocation of the potential was ipsilateral. In evoking the new instrument, we used the same parameters for the test, except only for filtering, which was 5 to 2,200 Hz filters used were different, since the gold-standard equipment, similar to equipment to capture auditory potentials available in the market, has a limitation to this setting. Thus, as the new device features a large number of settings, we used the most suitable filter configuration according to the literature.
To complete the interpretation of the findings, the responses were analyzed by morphology, standing out from the waves p13 and n23, the latencies of the first negative and positive peaks. Then we obtained the asymmetry index (13, 14), calculated by equation 1, which may be recalled the following:
R (%) = (AE - AD / AE + AD) .100 (1)
Where A = amplitude of VEMP and D and E in reference to the right and left sides, respectively.
Data were tabulated and processed by microcomputer application for Statistical Package for Social Sciences (SPSS) version 16.0. For a description of the data, was made use of graphical and tabular presentation of averages, medians, standard deviations and percentiles. The normality of the samples was observed by the Kolmogorov-Smirnov and Shapiro-Wilk.
After the data were analyzed with the use of descriptive statistics was applied to test non-parametric Wilcoxon test for comparison of latencies of components of the VEMP (p13 and n23), evoked by the two devices. The chi-square test was applied for comparison of frequencies obtained by calculating the asymmetry index. Values were considered significant at p less than 0.05 (p <0.05). The value of the beta error was 0.1.
RESULTS The sample consisted of 11 volunteers, 5 males (45.45%) and 6 female (54.55%). There was aged between 19 and 31 years, mean age of 21.18 years and standard deviation of 3.37 years.
The normality of the samples, as the latencies and amplitudes of waves p13 and n23, was observed using the Shapiro-Wilk and Kolmogorov-Smirnov test, however, there was no normal distribution of variables: amplitude of p13 and n23 right side, new equipment, and n23 amplitude on the left, towards the set the gold standard. Thus, we used nonparametric tests ofWilcoxon and Chi-square, depending on variables are averages or frequencies, respectively.
In all subjects, were captured waves p13 and n23 VEMP with appropriate morphology, by unilateral stimulation and capture, regardless of the device.
Comparing the two devices under study, without differentiating between right and left sides, it appears that absolute latencies later for the gold-standard equipment (Table 1).
Table 2 can be verified largest absolute amplitude (in module) for waves p13 and n23, evoked in the new instrument compared to the EP25.
The results, as the latencies and amplitudes of the average values between the left and right ears for the two instruments can be better visualized in Figure 1.
Applying the Wilcoxon test, we could confirm the absence of statistically significant differences when comparing the two instruments regarding the parameters of latency and amplitude components of the p13 and n23, except for the latency of n23, which we found a significant difference p-value = 0.005.
Making use of the formula for calculating the asymmetry index (13, 14) in normal subjects, we can observe higher values of asymmetry for the gold-standard unit (22.6%) than for the new instrument (17.3% ), as regards the p13 component. As the n23 component, these rates were higher in the recall by new equipment (10.1%), compared to 8.1% of the gold standard.
The chi-square, however, revealed no statistically significant differences between the new equipment market and the gold standard (EP25), when comparing the asymmetry indexes of the amplitudes of p13 and n23, with values of 0.14 and p = 0.60, respectively.
In this study, the value of asymmetry index inter-p13-n23 peak for the new unit (20.1%) was significantly lower than that found for the EP25 (30.5%), p less than 0.01 , also using the chi-square.
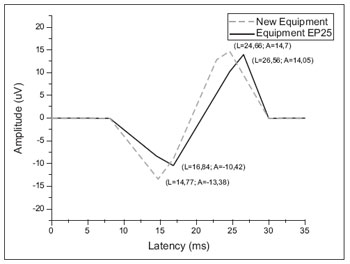
Figure 1. Mean values for latency and amplitude components of evoked VEMP in new equipment and gold apparatus standard.
They chose to tone burst stimuli at a frequency of 500 Hz, since they are more effective than clicks, to get the VEMP, and among the tone burst stimuli, the low frequencies (<1000 Hz) are more effective than high (4, 14), the 500 Hz, the most widely used (7, 14).
As the stimulus intensity was chosen dBNAn 90, since the majority of studies, using stimulus intensities at or above this intensity (15-18).
Been reported for several muscles VEMP recording (17, 19-21). In this study, we used the SCM muscle, as is currently the most used (14, 16, 22). Added to this, comparisons of results of VEMP in the SCM muscle and the trapezius muscle, show that the answers are more homogenous in the ECM (13).
Regarding the nomenclature of the waves of VEMP, the first peak may have the following designations: A, p13, p14 and I. The second peak is referred to in literature as n23, n21 or II (7, 24).In this study, we chose to use the classifications for the first and second peaks, respectively, p13 and n23, since they are normally used by the literature. The difference between the latencies and amplitudes between n23 and p13 was called inter-peak, confirming the expression of electrical activity in muscle reflex to sound stimulation of the saccular macula.
In this study, we analyzed only the waves p13 and n23, since the later waves are not present in all normal subjects and therefore should not be taken into account when interpreting VEMP (13).
Observation of the absolute latency of responses is important for the identification of peaks in the tracing and observation of problems that interfere with nerve conduction velocity (13), and is a reproducible parameter of VEMP response, independent of stimulus intensity and the voltage level electromyographic (1).
A study of 64 normal subjects (22), using the same methodology to test for evoking VEMP were determined normal values for latency of wave p13, up to 20.3 ms and 28.0 ms for n23. However, in another study (1), normal values for the p13 wave, varying between 13.8 and 18.5 ms, while, n23 shows variation from 20.6 to 27.4 ms. Thus, regardless of normal parameters and equipment used (the gold standard of the market or new device developed), the absolute latencies of the two components of the VEMP, were according to normal standards. However, it took longer time of registration to be received, each of the two waves, the gold standard in the appliance.
The results found in this work show that, although there are no statistically significant differences regarding the absolute latency between the two instruments can be found earlier latency values in the new instrument, probably resulting from the use of filters more particular, to capture this potential.
Thus, the absolute latencies clinical parameters are considered useful for the assessment of neural conduction, helping assist in the diagnosis of neurological disorders such as multiple sclerosis, for example (13).
The absolute values of amplitude, in this study were quite variable among individuals, and in the same subject, when captured at different times, both equipments (the gold standard and the new instrument). These findings agree with the majority of studies on VEMP (7, 22, 25-27).
With the evocation of VEMP on both devices, were found higher values of absolute amplitude in the new instrument. These findings configure quantitative increase in the activity of vestibular-cervical reflex (28). In addition, values of standard deviations were often smaller in the new unit, which portrays the variations decrease in amplitude of VEMP, evoked by this instrument.
Nevertheless, numerous authors agree that the absolute amplitude values should not be used in the analysis of this test, since it cannot be reproduced, due to the large intersubject variation, and are dependent on factors such as stimulus intensity and level tonic contraction of SCM muscle (7, 22, 25-28).
Furthermore, we observed absolute values of amplitude, for both devices, very different from that found in some studies that used the same test methodology, which can be explained by the use of different amplifier gains. Thus, the gain would be an influencing factor for the amplitude, which reinforces even more the non-use of absolute amplitude, the analysis of the VEMP.
In an attempt to counteract these effects become interfering and an amplitude parameter analyzable in VEMP test was performed using the asymmetry index (13, 14). Thus, through this equation, we can see normal results for the components p13 and n23, the second a previous study (14), which reports that they must be below 34%. Furthermore, it was proven, statistically, the absence of asymmetries for these waves, captured in the two rigs.
Another analysis of the VEMP was performed in the present study, considering the asymmetry index of the inter-peak amplitude p13-n23, showed much lower rates for the new unit (20.1%) than for the gold standard instrument (30, 5%) and also found in the previous study, 45.00% (13). This shows that the developed device has higher effectiveness in capturing the VEMP the industry standard.
Thus, for the inter-personal comparison of the magnitude of the responses should not be used the absolute values of these amplitudes, but the asymmetry index, since it reflects the interaural amplitude difference, weighted by the average amplitude of this response.
CONCLUSION There are no differences for the latencies and amplitudes of VEMP between the EP25 and the new device developed by presenting the latter, closer to the values reported in literature and a lower index of asymmetry of the inter-peak amplitude p13-n23.
BIBLIOGRAPHIC REFERENCES1. Damen MMJ. Vestibular evoked myogenic potential (VEMP). Clinical application of the threshold. Medical Engineering. Eindhoven: Technische Universiteit Eindhoven; 2007.
2. Rauch RD. Vestibular evoked myogenic potencials. Otol Neurotol. 2006, 14:299-304.
3. Sazgar AA, Akrami K, Akrami S, Yazdi ARK. Recording of vestibular evoked myogenic potencials. Acta Med Iran. 2006, 44(1):13-6.
4. Akin F, Murnane O, Panus P, Caruthers S, Wilkinson A, Proffitt T. The influence of voluntary tonic EMG level on the vestibular-evoked myogenic potencial. JRRD. 2004, 41(3B):473-80.
5. Sheykholeslami K, Kaga K. The otolithic organ as a receptor of vestibular hearing revealed by vestibular-evoked myogenic potentials in patients with inner ear anomalies. Hear Res. 2002, 165:62-7.
6. Rosengren SM, Colebatch JG. Vestibular evoked potencials (VsEPs) in patients with severe to profund bilateral hearing loss. Clin Neuroph. 2006, 117:1145-53.
7. Hall J. New handbook of auditory evoked responses. New York: Pearson Education; 2006.
8. Guillén VP, García EG, Piñero AG, Rey APD, Pérez CM. Potencial vestibular miogénico evocado: un aporte al conocimiento de la fisiología y patología vestibular: Patrones cuantitativos en la población normal. Acta Otorrinolaringol Esp. 2005, 56:349-53.
9. Sazgar A, Dortjai V, Akrami K, Akrami S, Yazdi AK. Saccular damage in patients with high-frequency sensorioneural hearing loss. Eur Arch Otorhinolaryngol. 2006, 263:608-13.
10. Takeich N, Sakamoto T, Fukuda S, Inuyama Y. Vestibular evoked myogenic potencial (VEMP) in patients with acoustic neuromas. Auris Nasus Lar. 2001, 28:839-41.
11. Pollak L, Kushnir M, Stryjer R. Diagnostic value of vestibular evoked myogenic potencials in cerebellar and lower-brainstem stroke. Neuroph Clin. 2006, 36:227-33.
12. Young Y. Vestibular-evoked myogenic potencials: optimal stimulation and clinical application. Jour of Biom Sci. 2006, 13:745-51.
13. Almeida RR. Potenciais evocados miogênicos vestibulares: Estudo em indivíduos normais. São Paulo: Universidade de São Paulo; 1999.
14. Murofushi T, Matsuzaki M, Wu C. Short tone burst-evoked myogenic potencials on the sternocleidomastoid muscle: are these potentials also of vestibular origin?. Arch Otolaryngol Head Neck Surg. 1999, 125:660-4.
15. Magliulo G, Cuiuli G, Gagliardi M, Ciniglio-Appiani G, DAmico R. Vestibular evoked myogenic potentials ang glycerol testing. Laryngoscope. 2004, 114:338-42.
16. Colebatch JG, Halmagyi GM, Skuse NF. Myogenic potencials generated by a click-evoked vestibulocollic reflex. Neurol Neurosurg. Psychiatry. 1994, 57:190-7.
17. Ferber-Viart C, Duclaux R, Colleaux B, Dubreuil C. Myogenic vestibular-evoked potentials in normal subjects: A comparison between responses obtained from sternomastoid and trapezius muscles. Acta Otolaryngol (Stockh). 1997, 117:472-81.
18. Ribeiro S, Almeida RR, Caovilla HH, Ganança MM. Dos potenciais evocados miogênicos vestibulares nas orelhas comprometida e assintomática na Doença de Meniére unilateral. Rev Bras Otorrinolaringol. 2005, 77(1):60-6.
19. Wu C, Young Y, Murofushi T. Tone burst-evoked myogenic potencials in human neck flexor and extensor. Acta Otolaryngol (Stockh). 1999, 119:741-4.
20. Sakakura K, Takahashi K, Takayasu Y, Chikamatsu K, Furuya N. Novel method for recording vestibular evoked myogenic potential: Minimally invasive recording on neck extensor muscles. Laryngoscope. 2005, 115:1768-73.
21. Cody D, Bickford R. Averaged evoked myogenic responses in normal man Laryngoscope. 1969, 79:400-16.
22. Basta D, Todt I, Ernst A. Normative data for P1/N1 - latencies of vestibular evoked myogenic potencials induced by air- or bone-conducted tone bursts. Clin Neuroph. 2005, 116:2216-9.
23. Welgampola M, Colebatch J. Characteristics and clinical applications of vestibular-evoked myogenic potencials. Neurol. 2005, 64:1682-8.
24. Murofushi T, Iwasaki S, Ozeki H, Ushio M, Chihara Y. Tone burst-galvanic ratio of vestibular evoked myogenic potential amplitudes: A new parameter of vestibular evoked myogenic potential? Clin Neuroph. 2007, 118:1685-90.
25. Ochi K, Ohashi T. Age-related changes in the vestibular-evoked myogenic potencials. Otolaryngol Head Neck Surg. 2003, 129:655-9.
26. Su H, Huang T, Young Y, Cheng P. Aging effect on vestibular evoked myogenic potencial. Otol Neurotol. 2004, 25:977-80.
27. Ochi K, Ohashi T, Nishino H. Variance of vestibular-evoked myogenic potencials. Laryngoscope. 2001, 111:522-7.
28. Lim CL, Clouston P, Sheean G, Yiannikas C. The influency of voluntary EMG activity and click evoked myogenic potencial. Muscle Nerve. 1995, 18:1210-3.
1 PhD in Medical Sciences, Faculty of Medicine of Ribeirao Preto - USP. Professor, Federal University of Sergipe.
2 Doctor of Medical Sciences, Otorhinolaryngology. Full Professor, Faculty of Medicine of Ribeirao Preto - USP.
3 Ph.D. in Applied Physics in Medicine and Biology. Adjunct Professor, State University of Health Sciences of Alagoas.
Institution: Department of Ophthalmology, Otolaryngology and Head and Neck. Faculty of Medicine of Ribeirao Preto - University of São Paulo (USP) and Laboratory Instrumentation and Acoustics (LIA) State University of Health Sciences of Alagoas (UNCISAL) Maceió / AL - Brazil. Correspondence to: Prof. Dr. Pedro de Lemos Menezes - Rua Dr. Antonio Cansação, 55 - Apt. 703 - Ponta Verde - Maceió / AL - Brazil - Zip code: 57035-190 - Telephone: (+55 82) 3315-6813 - E-mail: pedrodelemosmenezes@gmail.com
Article received on July 11, 2010. Approved on August 22, 2010.